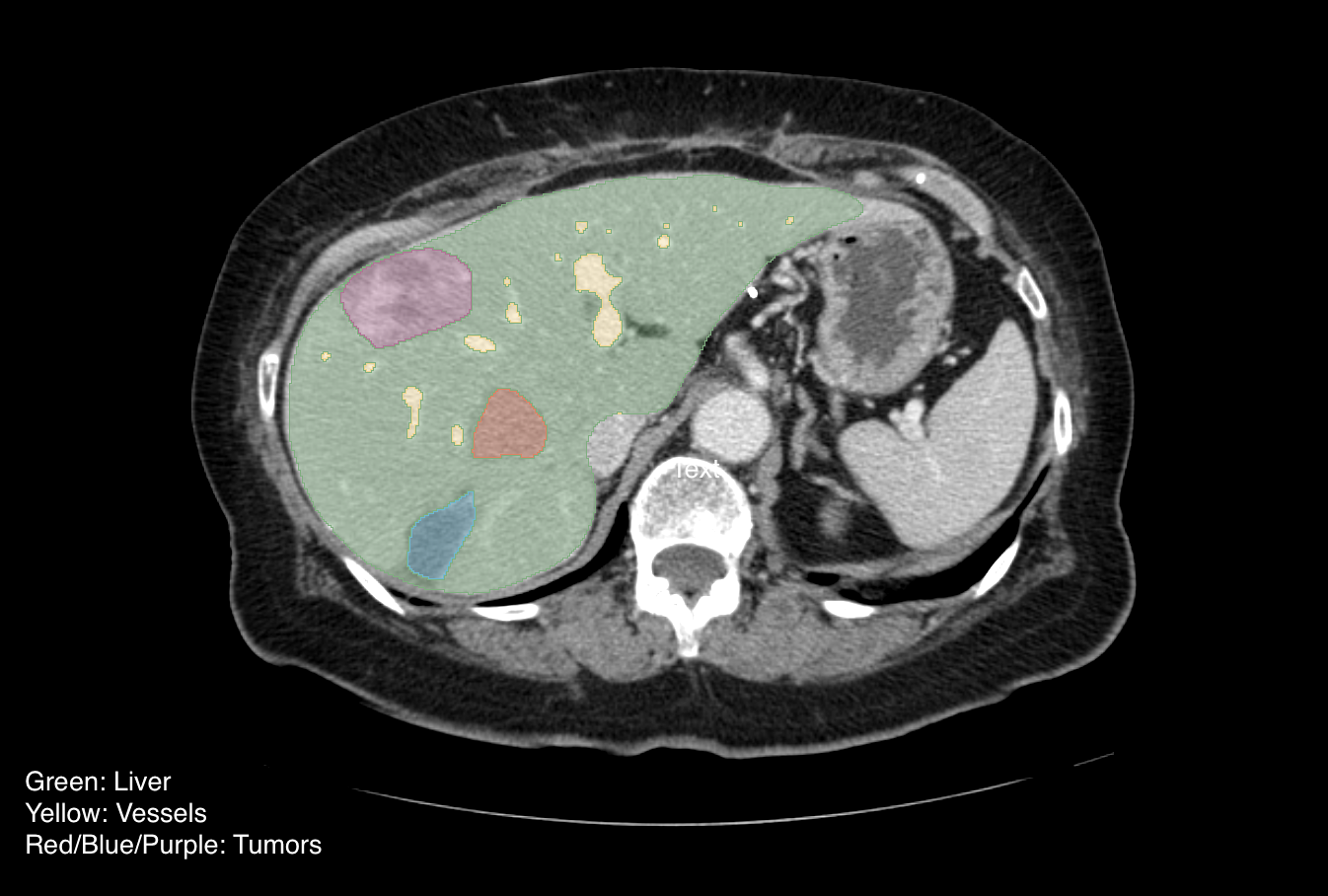
Colorectal-Liver-Metastases | Preoperative CT and Survival Data for Patients Undergoing Resection of Colorectal Liver Metastases
DOI: 10.7937/QXK2-QG03 | Data Citation Required | Image Collection
| Location | Species | Subjects | Data Types | Cancer Types | Size | Status | Updated | |
|---|---|---|---|---|---|---|---|---|
| Colon | Human | 197 | SEG, CT | Colorectal Cancer | Clinical | Public, Complete | 2023/04/25 |
Summary
This collection consists of DICOM images and DICOM Segmentation Objects (DSOs) for 197 patients with Colorectal Liver Metastases (CRLM). The collection consists of a large, single-institution consecutive series of patients that underwent resection of CRLM and matched preoperative computed tomography (CT) scans for quantitative image analysis. Inclusion criteria were (a) pathologically confirmed resected CRLM, (b) available data from pathologic analysis of the underlying non-tumoral liver parenchyma and hepatic tumor, (c) available preoperative conventional portal venous contrast-enhanced multi-detector computed tomography (MDCT) performed within 6 weeks of hepatic resection. Patients with 90-day mortality or that had less than 24 months of follow-up were excluded. Additionally, because pathologic and radiographic alterations of the non-tumoral liver parenchyma caused by hepatic artery infusion (HAI) of chemotherapy are not well described, any patient who received preoperative HAI was excluded. Finally, to obtain the most accurate future liver remnant (FLR), patients who underwent either local tumor ablation, more than 3 wedge resections, or had no visible tumor on preoperative imaging were excluded. The corresponding clinical dataset provides the variables collected including demographic, pathologic and survival data along with a data dictionary. The CT images were extracted from PACS and de-identified via institutional approved and a HIPAA compliant method. The liver, tumors, and vessels were semi-automatically segmented and a 3D model was generated using Scout Liver (Pathfinder Technologies Inc., TN, USA). Post-operative imaging and/or the resection margin width from pathology analysis were used to determine the transection lines needed to generate the segmentation of the liver remnant after surgery. Our informatics team converted the native MHD segmentation files into DICOM-SEG format using the open-source dcmqi software. The segments in a given DICOM Segmentation Object (DSO) can be consistently identified using the Segment Label header, which follows the naming scheme described below. The SCT codes used to categorize the segments in the Segmented Property Type Code Sequence header may be updated in future releases to comply with best practices. Full details regarding the data collection and preparation process can be found in our data descriptor paper: Simpson, A. L., Peoples, J., Creasy, J. M., Fichtinger, G., Gangai, N., Keshavamurthy, K. N., Lasso, A., Shia, J., D’Angelica, M. I., & Do, R. K. G. (2024). Preoperative CT and survival data for patients undergoing resection of colorectal liver metastases. In Scientific Data (Vol. 11, Issue 1). Springer Science and Business Media LLC. https://doi.org/10.1038/s41597-024-02981-2 Dataset: The dataset inside a subject folder is organized as follows: The DSOs are verified for all cases. Case "CRLM-CT-1074" was scanned with the subject in a prone (laying on stomach) position, so the images are in a different orientation than usual.
Data Access
Version 2: Updated 2023/04/25
Note: The clinical data was updated to provide an additional eight variables, in order to better match the description of the dataset in the original 2017 publication. The variables are: body_mass_index, node_positive_primary, synchronous_crlm, multiple_metastases, carcinoembryonic_antigen, max_tumor_size, bilobar_disease, preoperative_pve.
| Title | Data Type | Format | Access Points | Subjects | License | |||
|---|---|---|---|---|---|---|---|---|
| Images, Segmentations | SEG, CT | DICOM | Download requires NBIA Data Retriever |
197 | 197 | 394 | 17,836 | CC BY 4.0 |
| Clinical data | CSV | CC BY 4.0 |
Additional Resources for this Dataset
The NCI Cancer Research Data Commons (CRDC) provides access to additional data and a cloud-based data science infrastructure that connects data sets with analytics tools to allow users to share, integrate, analyze, and visualize cancer research data.
- Imaging Data Commons (IDC) (Imaging Data)
Citations & Data Usage Policy
Data Citation Required: Users must abide by the TCIA Data Usage Policy and Restrictions. Attribution must include the following citation, including the Digital Object Identifier:
Data Citation |
|
|
Simpson, A. L., Peoples, J., Creasy, J. M., Fichtinger, G., Gangai, N., Lasso, A., Keshava Murthy, K. N., Shia, J., D’Angelica, M. I., & Do, R. K. G. (2023). Preoperative CT and Survival Data for Patients Undergoing Resection of Colorectal Liver Metastases (Colorectal-Liver-Metastases) (Version 2) [Data set]. The Cancer Imaging Archive. https://doi.org/10.7937/QXK2-QG03 |
Acknowledgements
This study was funded in part through the NIH/NCI Cancer Center Support Grant P30 CA008748 and NIH supported grant R01CA233888.
Related Publications
Publication Citation |
|
|
Simpson, A. L., Peoples, J., Creasy, J. M., Fichtinger, G., Gangai, N., Keshavamurthy, K. N., Lasso, A., Shia, J., D’Angelica, M. I., & Do, R. K. G. (2024). Preoperative CT and survival data for patients undergoing resection of colorectal liver metastases. In Scientific Data (Vol. 11, Issue 1). Springer Science and Business Media LLC. https://doi.org/10.1038/s41597-024-02981-2 |
Research Community Publications
TCIA maintains a list of publications which leverage TCIA data. If you have a manuscript you’d like to add please contact the TCIA Helpdesk.
Previous Versions
Version 1: Updated 2023/03/01
| Title | Data Type | Format | Access Points | License | ||||
|---|---|---|---|---|---|---|---|---|
| Images | DICOM | Download requires NBIA Data Retriever |
CC BY 4.0 | |||||
| Clinical Data | CSV | CC BY 4.0 |
